Regarding this, what is the electron configuration of Cu 2+?
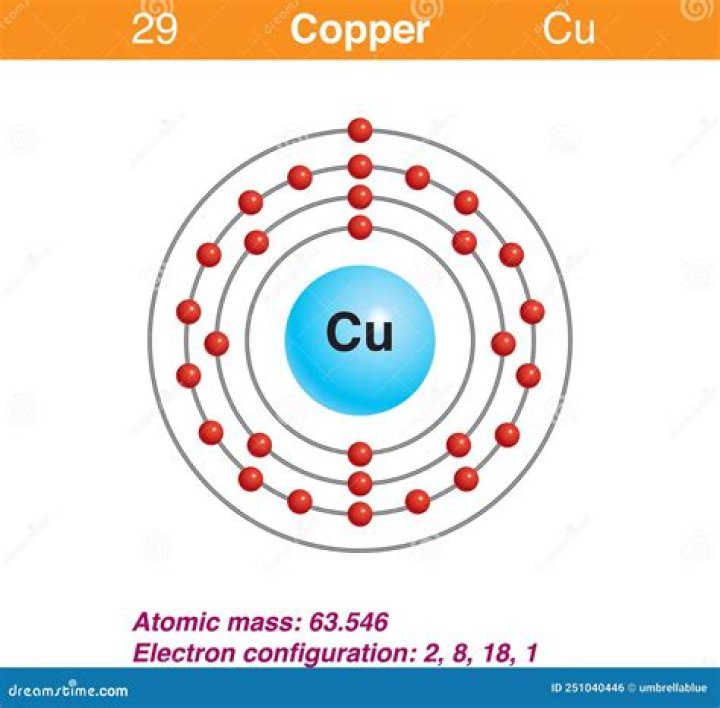
Electronic configuration of Cu is 1s2, 2s2, 2p6, 3s2, 3p6, 4s2, 3d9 ([Ar] 4s2, 3d9), whereas for Cu2+ is [Ar], 3d9.
Also Know, why is the electron configuration for copper 1s22s22p63s23p63d104s1 instead of 1s22s22p63s23p63d94s2? A filled sublevel is more stable than a half-filled sublevel. The arrangement of electrons with the same spin is as small as possible. The 4s orbital has higher energy than the 3d orbital.
Herein, why the electronic configuration of copper is?
It is due to full filled stability which the copper achieves when electron jump to d orbital to achieve full filled stability. 1. Exchange energy -in d orbital there are 5 subshell so ten exchanges of electron is possible which imparts much stability to copper hence this process is exothermic in nature.
Is CU 1 or CU 2 more stable?
Cu2+ is more stable than Cu+, Explain. Cu2+ is more stable than Cu+.Stability depends on the hydration energy (enthalpy) of the ions when they bond to the water molecules. The Cu2+ ion has a greater charge density than Cu+ ion and thus forms much stronger bonds releasing more energy.
Which is larger Cu+ or cu2+?
Cu+ is larger than Cu2+. Cu2+ has a smaller cationic radius due to stronger hold of the nucleus on the remaining 27 electrons after the loss of two as compared to Cu+ where only one electron is lost and the hold of nucleus is less.What is the difference between Cu and cu2+?
---> Cu is a neutral atom where number of protons and electrons are equal. ----> Cu2+ is a cation formed by lossing 2 electrons. As proton number is more, nuclear Charge for Cu²? will be more and atomic size of Cu²? decreases, Ionization potential is more. So atomic radius of Cu atom is more than Cu2+ ion.Which ion is more stable fe2+ or fe3+?
Hello yaar, Fe3+ ion is more stable due to its half-filled 3d5 electron configuration. As half filled and completely filled shells are more stable Fe3+ ion is more stable. While Fe2+ is not stable.What is cu2+ name?
The old style system has different suffixes in their names. For example, Fe2+ is called the ferrous ion, and Fe3+ is called the ferric ion; Cu+ is the cuprous ion, and Cu2+ is the cupric ion.Why cu2+ is more stable than Cu+?
Stability depends on the hydration energy (enthalpy) of the ions when they bond to the water molecules. The Cu2+ ion has a greater charge density than Cu+ ion and thus forms much stronger bonds releasing more energy.What is the magnetic moment of cu2+?
The magnetic moment of Cu+2 is root 3. It has 2 unpaired electron. Root of no (n+2). Therefore root 3,as the value of n for Cu+2 is 2.What is the electron configuration of Cu 1?
Copper is found in two common ionic forms, Cuprous Cu(I) with a noble gas electron configuration of [Ar]4s03d10 and Cupric Cu(II) with [Ar]4s03d9.How many electrons are in each shell?
Each shell can contain only a fixed number of electrons: The first shell can hold up to two electrons, the second shell can hold up to eight (2 + 6) electrons, the third shell can hold up to 18 (2 + 6 + 10) and so on. The general formula is that the nth shell can in principle hold up to 2(n2) electrons.What element has the electron configuration of 1s2 2s2 2p6 3s2?
Electron Configuration Match 1--Complete Address| A | B |
|---|---|
| neon | 1s2 2s2 2p6 |
| sodium | 1s2 2s2 2p6 3s1 |
| magnesium | 1s2 2s2 2p6 3s2 |
| aluminum | 1s2 2s2 2p6 3s2 3p1 |