| Number of Energy Levels: | 4 |
|---|---|
| First Energy Level: | 2 |
| Second Energy Level: | 8 |
| Third Energy Level: | 14 |
| Fourth Energy Level: | 2 |
In this regard, how many principal energy levels are there?
four
Likewise, how many orbitals are there in iron? When we write the configuration we'll put all 26 electrons in orbitals around the nucleus of the Iron atom. In writing the electron configuration for Iron the first two electrons will go in the 1s orbital.
People also ask, why is 3rd shell 8 or 18?
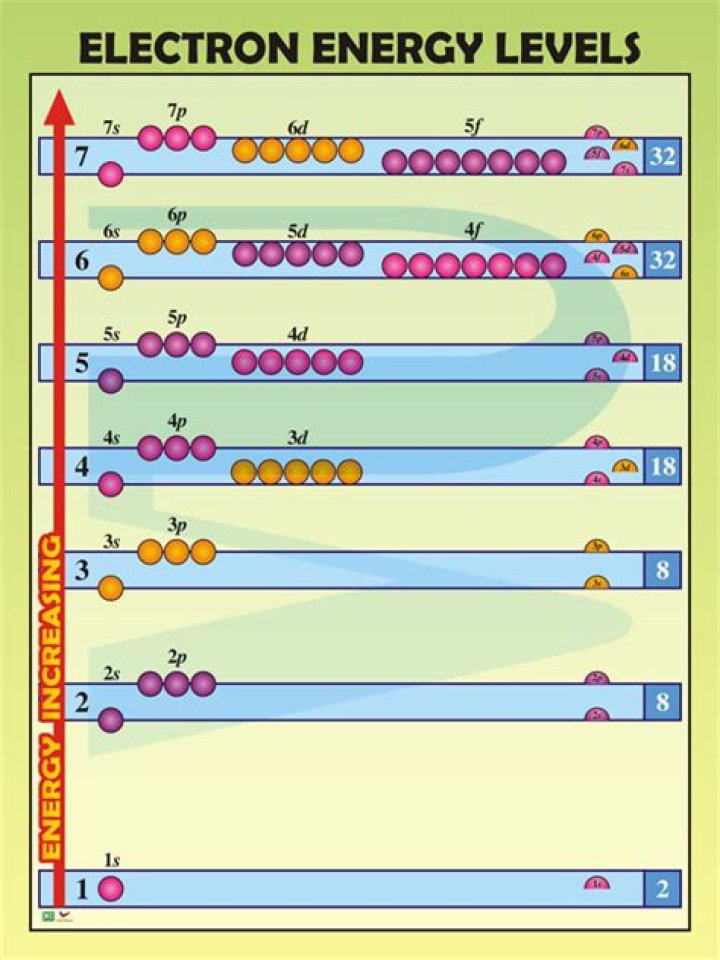
Each shell can contain only a fixed number of electrons: The first shell can hold up to two electrons, the second shell can hold up to eight (2 + 6) electrons, the third shell can hold up to 18 (2 + 6 + 10) and so on. The general formula is that the nth shell can in principle hold up to 2(n2) electrons.
What is the highest principal energy level?
Sodium is in column 1 of the third period. In a sodium atom, the highest-energy principal energy level containing electrons is the third energy level, and that energy level contains one electron.
How are energy levels calculated?
The formula defining the energy levels of a Hydrogen atom are given by the equation: E = -E0/n2, where E0 = 13.6 eV (1 eV = 1.602×10-19 Joules) and n = 1,2,3… and so on. The energy is expressed as a negative number because it takes that much energy to unbind (ionize) the electron from the nucleus.What is the symbol for principal energy level?
D. In chemistry, the principal energy level of an electron refers to the shell or orbital in which the electron is located relative to the atom's nucleus. This level is denoted by the principal quantum number n.What does highest energy level mean?
It is the highest-energy atomic orbital in an atom that is filled with electrons. Ordering orbitals by energy is straightforward; energy is quantized, so the higher the principal quantum number n , usually the higher the energy of the orbital.What is Hund rule?
Hund's Rule. Hund's rule: every orbital in a subshell is singly occupied with one electron before any one orbital is doubly occupied, and all electrons in singly occupied orbitals have the same spin.Which orbitals have the highest energy?
The 1s orbital has the highest energy. You can understand it by thinking about different things: But first let's be super clear: the energy of an electron is the energy it would require to rip it out of the atom's electronic cloud.What are the 4 types of sublevels?
Level 4 has 4 sublevels - s, p, d, and f. These are pictured below. The sublevels contain orbitals. Orbitals are spaces that have a high probability of containing an electron.What is meant by energy level?
Energy levels inside an atom are the specific energies that electrons can have when occupying specific orbitals. Electrons can be excited to higher energy levels by absorbing energy from the surroundings. Light is emitted when an electron relaxes from a high energy state to a lower one.How is Valency calculated?
The valency of an atom is equal to the number of electrons in the outer shell if that number is four or less. Otherwise, the valency is equal to eight minus the number of electrons in the outer shell. Once you know the number of electrons, you can easily calculate the valency.Why does the third period have 8 elements?
According to the Aufbau's Principle (the building up principle) there can be only EIGHT elements that satisfy this. This is because the S orbital of the fourth shell sits at a lower energy level (closer to the nucleus) than the d orbital of the third shell.What is the capacity of M shell?
Questions and Answers| Energy Level (Principal Quantum Number) | Shell Letter | Electron Capacity |
|---|---|---|
| 1 | K | 2 |
| 2 | L | 8 |
| 3 | M | 18 |
| 4 | N | 32 |
How many 2s electrons are in Li?
Therefore its atoms contain 3 protons. A neutral atom has equal numbers of protons and electrons, so a neutral Li atom also has 3 electrons. The electron configuration of Li is 1s22s1 . So you can see that there are 2 inner electrons in the 1s sublevel.How many electrons can fit in the 4th Shell?
The fourth energy level has 18 electrons. The fourth energy level of the periodic table includes the 4s 3d and 4p orbitals. The 4p orbital holds 6 electrons.What is the 2 8 8 rule in chemistry?
The 2–8–8 rule is the electron filling rule in the shells of an atom. It is used for predicting the no. Of electron in each shell. The innermost shell will have maximum of 2 electrons, second will have 8 and so on. It follows a rule of 2n^2, where n is equal to the position of shell .Why do shells start with K?
He noticed that atoms appeared to emit two types of X-rays. As it turns out, the K type X-ray is the highest energy X-ray an atom can emit. It is produced when an electron in the innermost shell is knocked free and then recaptured. This innermost shell is now called the K-shell, after the label used for the X-ray.What is Subshell?
A subshell is a subdivision of electron shells separated by electron orbitals. Subshells are labelled s, p, d, and f in an electron configuration.What are the sublevels?
A sublevel is an energy level defined by quantum theory. In chemistry, sublevels refer to energies associated with electrons. In physics, sublevels may also refer to energies associated with the nucleus. Shell 4 can hold up to 32 electrons, Shell 5 can hold up to 50 electrons,How do you find the principal energy level of an element?
Number of energy levels in each period- The atoms in the first period have electrons in 1 energy level.
- The atoms in the second period have electrons in 2 energy levels.
- The atoms in the third period have electrons in 3 energy levels.
- The atoms in the fourth period have electrons in 4 energy levels.