Number of energy levels in each period The atoms in the first period have electrons in 1 energy level. The atoms in the second period have electrons in 2 energy levels. The atoms in the third period have electrons in 3 energy levels. The atoms in the fourth period have electrons in 4 energy levels.Also to know is, how do you find the energy level of an atom?
The formula defining the energy levels of a Hydrogen atom are given by the equation: E = -E0/n2, where E0 = 13.6 eV (1 eV = 1.602×10-19 Joules) and n = 1,2,3… and so on. The energy is expressed as a negative number because it takes that much energy to unbind (ionize) the electron from the nucleus.
Subsequently, question is, how many energy levels are occupied in an atom of oxygen?
| Element | Element Number | Number of Electrons in each Level |
| Oxygen | 8 | 6 |
| Fluorine | 9 | 7 |
| Neon | 10 | 8 |
| Sodium | 11 | 8 |
Correspondingly, how many electrons are in each energy level?
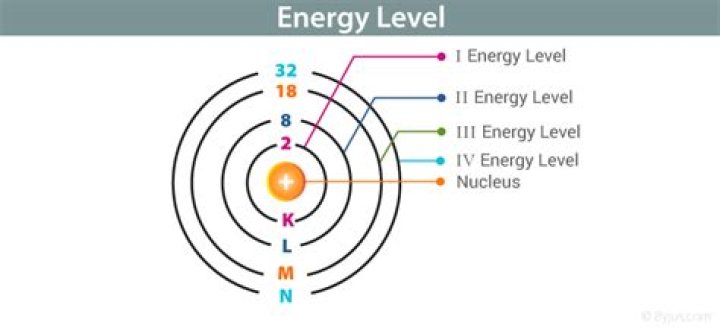
Each shell can contain only a fixed number of electrons: The first shell can hold up to two electrons, the second shell can hold up to eight (2 + 6) electrons, the third shell can hold up to 18 (2 + 6 + 10) and so on. The general formula is that the nth shell can in principle hold up to 2(n2) electrons.
Where is the lowest energy level in an atom?
The lowest level is sometimes referred to as the ground state. This is the 1s orbital, and has an energy debt of -13.6 eV in hydrogen. It varies from atom to atom depending on the number of protons in the nucleus. Each orbital only can support so many electrons, which are separated slightly by spin differences.
Do atoms have energy?
Atoms do not have to get energy from somewhere, because they are energy!! This might be hard to swallow, but its true. Einstein proposed that mass and energy are two sides of the same coin. Mass can convert into energy and vice-versa.Are atoms energy?
All matter is made up of atoms. These shells are actually different energy levels and within the energy levels, the electrons orbit the nucleus of the atom. The ground state of an electron, the energy level it normally occupies, is the state of lowest energy for that electron.How is energy stored in atoms?
Chemical energy is energy stored in the bonds of atoms and molecules. It is the energy that holds these particles together. Nuclear energy is energy stored in the nucleus of an atom; it is the energy that holds the nucleus together. The energy can be released when the nuclei are combined or split apart.What is Bohr equation?
Bohr equation. The Bohr equation, named after Danish physician Christian Bohr (1855–1911), describes the amount of physiological dead space in a person's lungs. This is given as a ratio of dead space to tidal volume. It differs from anatomical dead space as measured by Fowler's method as it includes alveolar dead spaceWhat is meant by energy level?
Energy levels inside an atom are the specific energies that electrons can have when occupying specific orbitals. Electrons can be excited to higher energy levels by absorbing energy from the surroundings. Light is emitted when an electron relaxes from a high energy state to a lower one.What are the main energy levels?
A principal energy level may contain up to 2n2 electrons, with n being the number of each level. The first energy level can contain 2(1)2 or two electrons; the second can contain up to 2(2)2 or eight electrons; the third can contain up to 2(3)2 or 18 electrons, and so on.What are sublevels?
A sublevel is an energy level defined by quantum theory. In chemistry, sublevels refer to energies associated with electrons. In physics, sublevels may also refer to energies associated with the nucleus. Shell 4 can hold up to 32 electrons, Shell 5 can hold up to 50 electrons,How is Valency calculated?
The valency of an atom is equal to the number of electrons in the outer shell if that number is four or less. Otherwise, the valency is equal to eight minus the number of electrons in the outer shell. Once you know the number of electrons, you can easily calculate the valency.How many electrons are in the 2nd energy level?
eight electrons
What is a Subshell?
A subshell is a subdivision of electron shells separated by electron orbitals. Subshells are labelled s, p, d, and f in an electron configuration.Why are there only 2 electrons in the first shell?
This first shell has only one subshell (labeled 1s) and can hold a maximum of 2 electrons. This is why there are two elements in the first row of the periodic table (H & He). Because the first shell can only hold a maximum of 2 electrons, the third electron must go into the second shell.How many electrons are in the energy levels?
Each shell can contain only a fixed number of electrons: The first shell can hold up to two electrons, the second shell can hold up to eight (2 + 6) electrons, the third shell can hold up to 18 (2 + 6 + 10) and so on. The general formula is that the nth shell can in principle hold up to 2(n2) electrons.How many electrons are in the 7th energy level?
You can determine the number of orbitals you have in a particular shell by using the principal quantum number, n . Notice that you can easily find the number of electrons that an atom can accomodate on a particular shell, or energy level, by using the fact that each orbital can hold a maximum of two electrons.Why do shells start with K?
He noticed that atoms appeared to emit two types of X-rays. As it turns out, the K type X-ray is the highest energy X-ray an atom can emit. It is produced when an electron in the innermost shell is knocked free and then recaptured. This innermost shell is now called the K-shell, after the label used for the X-ray.How many electrons are in the 5th energy level?
The fifth shell has the s, p, d, f, and g subshells ? 2 + 6 + 10 + 14 + 18 = 50 electrons.What are the 4 types of sublevels?
Level 4 has 4 sublevels - s, p, d, and f. These are pictured below. The sublevels contain orbitals. Orbitals are spaces that have a high probability of containing an electron.What is Oxygen's atom called?
Oxygen is a chemical element – a substance that contains only one type of atom. Its official chemical symbol is O, and its atomic number is 8, which means that an oxygen atom has eight protons in its nucleus. Oxygen is normally found as a molecule. It is called dioxygen.