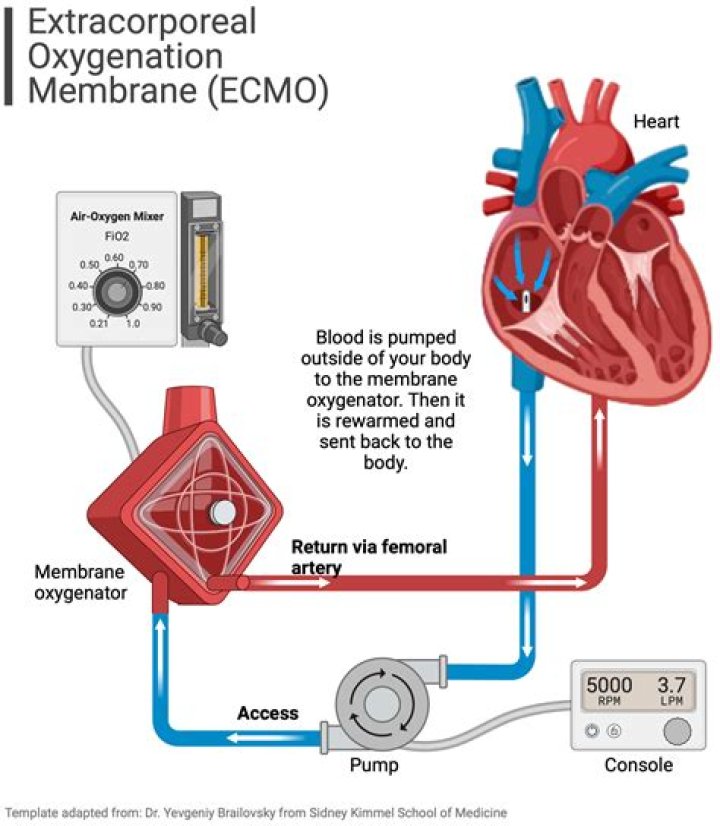
How does it work? The Impella Ventricular Support System helps pump blood by drawing blood out of the heart and pumping it into the aorta, partially or fully bypassing the left ventricle. It is implanted into the left side of a patient's heart through a small incision in the femoral artery (major artery in the leg).Also to know is, what is the Impella heart pump?
The world's smallest heart pump. Impella® is a heart pump intended for temporary use in patients with severe symptomatic CAD and diminished (but stable) heart function who are undergoing high-risk PCI.
Furthermore, what does the Impella do? Impella is a miniaturized percutaneously inserted ventricular assist device (VAD) that can be placed via a retrograde approach across the aortic valve using a femoral arterial access. The device pumps blood from the left ventricle into the ascending aorta and systemic circulation at the upper rate of 2.5 L/min.
Similarly, you may ask, how long can impella be used?
The FDA has approved use of Impella for 6 hours, but current data has revealed off-label use in patients for a variable duration, ranging from a few hours to an average of 12 days, with the longest recorded duration at 35 days in a single documented case.
Where does the Impella sit in the heart?
When properly positioned, the Impella Catheter delivers blood from the inlet area, which sits inside the left ventricle, through the cannula, to the outlet opening in the ascending aorta.
How much does an Impella cost?
Despite the fact that Impella devices are much more costly than balloon pumps, $22,000 (Impella 2.5) compared with $800-$1,000 (estimations), in terms of quality of life, Impella patients may fair better.What is the difference between IABP and impella?
The major difference between the two devices is that the IABP works by using the force of each heartbeat to increase blood flow while the Impella 2.5 device functions independently of how forceful your heart beats, pumping up to 2.5 liters of blood per minute. IABP has been used by cardiologists since the 1960s.How do I get rid of Impella?
Traditionally, the Impella® devices are removed via surgical repair of the common femoral artery, or percutaneously with applied pressure at the access site to achieve hemostasis. However, hemostasis can be difficult to obtain with pressure due to large sheath size.What is high risk PCI?
High-risk PCI was defined as the presence of impaired LV function (ejection fraction <30%) and extensive multivessel coronary disease, critical left main stenosis, or a target vessel that provides collateral supply to an occluded second vessel that in turn supplies >40% of myocardium.How long can a balloon pump stay in?
Prolonged use for at least 10 days of intraaortic balloon pumping (IABP) for heart failure.What does Impella CP mean?
Impella CP is a member of the Impella family of heart pumps which have the unique ability to unload the heart and enable. native heart recovery, potentially allowing patients to return home with their own hearts.What is a PCI procedure?
Percutaneous Coronary Intervention (PCI, formerly known as angioplasty with stent) is a non-surgical procedure that uses a catheter (a thin flexible tube) to place a small structure called a stent to open up blood vessels in the heart that have been narrowed by plaque buildup, a condition known as atherosclerosis.What is a tandem heart?
The TandemHeart is a left atrial-to-femoral artery bypass system that includes a transseptal cannula, arterial cannulae, and a centrifugal blood pump (Fig. Blood is then pumped into the femoral artery through the 15F or 17F arterial cannulae.Is an Impella permanent?
Visit to see a video showing insertion of the Impella 2.5 catheter. During your procedure, the Impella will be permanently connected to the console, which will be monitored by your caregivers.Can you do CPR on a patient with an Impella?
Cardiopulmonary support (CPR) should be initiated immediately per hospital protocol if indicated for any patient supported with the Impella® RP Catheter. During defibrillation, do NOT touch the Impella® RP Catheter, cables, or Automated Impella® Controller.Is impella FDA approved?
The Impella 2.5® and Impella CP® devices are U.S. FDA PMA approved to treat certain advanced heart failure patients undergoing elective and urgent percutaneous coronary interventions (PCI) such as stenting or balloon angioplasty, to re-open blocked coronary arteries.Where is the Impella placed?
The Impella CP device can be inserted via a standard catheterization procedure through the femoral artery, into the ascending aorta, across the valve and into the left ventricle. Impella CP® heart pump is safe and effective for use during high-risk PCI procedures and for patients in cardiogenic shock.What is purge pressure impella?
To prevent blood from entering the Impella® motor, a pressure barrier is created by running a purge solution. through the Impella® Catheter in the opposite direction of blood being drawn into the catheter. A recommended 300 mmHg of purge pressure is maintained by running a dextrose solution through the system.How is the Impella 5.0 catheter inserted?
Insertion: The Impella 5.0 can be inserted into the left ventricle via femoral cut down or through the axillary artery, and goes through the ascending aorta, across the valve and into the left ventricle.What is hemodynamic support?
Hemodynamic support in septic shock is provided by restoring the adequate circulating blood volume, and, if necessary, optimizing perfusion pressure and cardiac function with vasoactive and inotropic support to improve tissue oxygenation.How long can a person live with a LVAD?
Among those 18 patients, the median time from LVAD implantation to death was 16 days (range 1–270 days), with 5 dying within a week of implantation.What is cardiogenic shock?
Cardiogenic shock is a condition in which your heart suddenly can't pump enough blood to meet your body's needs. The condition is most often caused by a severe heart attack, but not everyone who has a heart attack has cardiogenic shock. Cardiogenic shock is rare, but it's often fatal if not treated immediately.